HEALTH CARE BRIEFING: US Gets First Plane of Formula From Europe
Bloomberg Government subscribers get the stories like this first. Act now and gain unlimited access to everything you need to know. Learn more.
A planeload of more than 70,000 pounds of baby formula arrived in the US, starting an emergency program to alleviate a national shortage that has left some parents scrounging to feed their children. More formula will start arriving in stores “as early as this week,” Brian Deese, President Joe Biden’s top economic adviser, said on CNN.
The plane that landed in Indianapolis on Sunday will address “15% of the overall national volume that we need,” he said.
Faced with pressure to address the crisis, Biden last week ordered the use of federal government planes to airlift infant formula. He also invoked emergency authority under the Defense Production Act to spur domestic manufacturing.
The US Air Force cargo plane from Ramstein Air Base in Germany delivered 132 pallets of Nestlé Health Science Alfamino Infant and Alfamino Junior formula. A second flight of Nestlé formula for children with cow’s milk protein allergy will depart Ramstein for Pennsylvania in the coming days, the White House said in a statement.
Deese said the shortage highlights how few companies are making baby formula. “How did we end up in a market where we have three companies that control 90% of the market?” he said. Read more from Victoria Cavaliere.
- Social media posts containing dangerous misinformation about homemade formula recipes have gone viral on the internet. Although major networks like Facebook, TikTok and YouTube have taken steps to label posts pointing to the harms of such recipes, and in some cases removed them, they have done so inconsistently, allowing the advice to continue spreading and putting children at risk. Read more from Davey Alba.
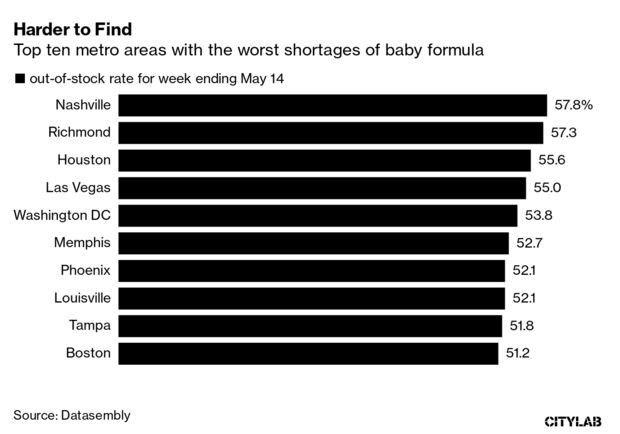
- The number of US cities with severe baby formula shortages grew over the week ending on May 14. There are now 11 cities with out-of-stock rates above 50% for the week ending on May 14, up from eight the week prior. Read more from Martine Paris.
Happening on the Hill
Hearings This Week:
- Labor-HHS-Education Member Day: The House Appropriations Labor-HHS-Education Subcommittee holds a Tuesday hearing where members can discuss issues of importance to their constituents.
- Nominations: The Senate Health, Education, Labor and Pensions Committee holds a Wednesday hearing to consider Rita Landgraf’s nomination to be assistant HHS secretary for aging. The Senate Indian Affairs Committee also holds a Wednesday hearing to consider Roselyn Tso’s nomination to be director of the Indian Health Service at the HHS.
- Baby Formula Shortage: The House Energy and Commerce Oversight Subcommittee holds a Wednesday hearing on the nation’s baby formula shortage, with FDA Commissioner Robert Califf set to testify. Califf also plans to appear before the Senate Health, Education, Labor and Pensions Committee on the same issue Thursday. Earlier: Baby-Formula Executives to Face House Hearing on Shortages
- BGOV Tracker: For a complete list of health-related hearings, click here.
Democrats Push Senate Drug Pricing Action: Rep. Susan Wild (D-Pa.) and 19 others called on Senate leadership to take action to lower prescription drug prices through budget reconciliation, which would only require a simple majority for passage. They urged that the bill “include capping the cost of insulin at $35 per month and out of pocket costs for seniors in Medicare Part D at $2,000 per year,” adding “finally giving Medicare the authority to negotiate drug prices will put thousands of dollars back in the pockets of hardworking Americans.” Read their letter here.
Grassley Backs SuperValu Whistleblowers in SCOTUS Challenge: Sen. Chuck Grassley (R-Iowa) told the Supreme Court that an appeals court improperly rejected whistleblowers’ False Claims Act suit alleging that SuperValu overcharged federal healthcare programs for prescription drugs. Daniel Seiden has more.
Industry & Regulations
Monkeypox Shouldn’t Spur Covid-19 Level of Concern, Biden Says: Biden sought to reassure Americans that the current monkeypox outbreak was unlikely to cause a pandemic on the scale of Covid-19. “I just don’t think it rises to the level of the kind of concern that existed with Covid-19,” he told reporters Monday in Tokyo at a joint press conference with Japanese Prime Minister Fumio Kishida.
Read more from Nancy Cook and Jennifer Jacobs.
Califf Taps CDC Official to Lead FDA Tobacco Center: A top official in the CDC’s Office on Smoking and Health will soon lead the FDA’s tobacco center as it carries out its new regulatory authority over synthetic nicotine and proposed bans on menthol in cigarettes and cigars. FDA Commissioner Califf announced Friday that he selected Brian King, a deputy director at the Centers for Disease Control and Prevention, to serve as the new director for the FDA Center for Tobacco Products, Celine Castronuovo reports.
Medical Gas Makers to Get New Safety Reporting Rules: Manufacturers of medical gases like oxygen and carbon dioxide would have to report serious adverse events within 15 days under a new rule floated by the FDA on Friday. Celine Castronuovo has more.
From the Courts
Contaminated Vaccine Case Is Bogus, Emergent Says: Emergent BioSolutions is accusing disgruntled shareholders of concocting a phony securities fraud case over statements surrounding the company’s contamination of Covid-19 vaccines. The suit stems from findings that staff at a company facility mixed AstraZeneca vaccine components into a Johnson & Johnson vaccine batch. Ian Lopez has more.
Ky. 15-Week Abortion Ban to Stay Blocked Under Ruling: Kentucky’s new law banning abortions after 15 weeks and imposing administrative and reporting requirements on providers will remain blocked after a judge partially granted a preliminary injunction requested by the state’s two abortion providers. Read more from Jacklyn Wille.
Bid to Save Roe Comes Via ERA Suits: The threat of a Supreme Court ruling overturning Roe v. Wade has spurred a trio of new lawsuits seeking state court recognition of the Equal Rights Amendment—a last-minute effort to preempt that anti-Roe ruling by forcing the high court to factor new constitutional language into its decision. The women’s rights group Elizabeth Cady Stanton Trust filed the lawsuits. Read more from Chris Marr.
HHS Defends Medicare Overpayment Rule in High Court Row: A rule requiring Medicare Advantage private insurers to refund overpayments was properly reinstated, the Department of Health and Human Services said in a brief filed with the U.S. Supreme Court. Read more from Christopher Brown.
More Headlines:
- Biotech’s $419 Billion Selloff Suggests a Recovery Is Years Away
- ARcare Patient Sues Over Data Breach Affecting Nearly 350,000
To contact the reporter on this story: Brandon Lee in Washington at blee@bgov.com
To contact the editors responsible for this story: Giuseppe Macri at gmacri@bgov.com; Michaela Ross at mross@bgov.com; Andrew Small at asmall@bgov.com
Stay informed with more news like this – from the largest team of reporters on Capitol Hill – subscribe to Bloomberg Government today. Learn more.