HEALTH CARE BRIEFING: U.S. Probing Mask Mandate Bans in Schools
Bloomberg Government subscribers get the stories like this first. Act now and gain unlimited access to everything you need to know. Learn more.
The Department of Education opened civil rights investigations in five Republican-led states asking whether bans on school mask mandates discriminate against students with disabilities at severe risk from Covid-19. Its Office for Civil Rights sent letters to state education leaders in Iowa, Oklahoma, South Carolina, Tennessee, and Utah yesterday notifying them of the reviews.
“It’s simply unacceptable that state leaders are putting politics over the health and education of the students they took an oath to serve,” Secretary Miguel Cardona said in a statement announcing the probe. “The Department will fight to protect every student’s right to access in-person learning safely and the rights of local educators to put in place policies that allow all students to return to the classroom full-time in-person safely this fall.”
Cardona and President Joe Biden have prioritized reopening of K-12 schools for in-person instruction this fall after most campuses shut down last year to halt the spread of the virus. But a surge in cases driven by the delta variant and political fighting over measures like universal masking indoors threaten to upend that goal if transmission in schools disrupts classes again.
The Office for Civil Rights will act as a neutral fact finder in the investigations, the letters told state education leaders. The reviews will look at whether mask mandate bans violate Title II of the Americans with Disabilities Act and Section 504 of the Rehabilitation Act of 1973, which protects students with disabilities against discrimination.
The investigations don’t include Florida, Texas, Arkansas, or Arizona, where state leaders have attempted to impose bans on local mask mandates but have been stopped by court orders. The Education Department said it will watch whether those states seek to further prevent school masking requirements and whether those court decisions are overturned.
Parents in South Carolina, Florida, and Texas have filed complaints in federal court challenging the state bans on mask mandates in schools. The lawsuits in all three states say the policies violate federal disability protections. Multiple states and school districts have adopted vaccine requirements for teachers and staff, but minors under age 12 are not yet eligible for vaccination in the U.S. Read more from Andrew Kreighbaum and Courtney Rozen.
More on the Pandemic
CDC Panel Backs Pfizer Shot, Punts on Booster: Pfizer and BioNTech’s Covid-19 vaccine got the backing of key health advisers, putting it on the cusp of a final signoff that would help governments and business push wider adoption. The Centers for Disease Control and Prevention’s Advisory Committee on Immunization Practices voted unanimously in favor of the two-dose regimen in people 16 and older yesterday. It still needs a final nod from CDC Director Rochelle Walensky.
The Pfizer-BioNTech shot’s approval last week by the Food and Drug Administration is the first for a Covid-19 vaccine in the U.S., coming as the Biden administration tries to control the spread of the highly transmissible delta strain. The White House expects the approval to help address hesitancy by raising confidence among those who say they’re wary of the shot’s fast development. One-fifth of Americans who are still unvaccinated said they’d only get shots if required, data at the meeting showed.
Yesterday’s meeting was originally intended to be occupied with the discussion of boosters more broadly. Now it appears that the bulk of those discussions will take place in mid-September, as the White House rushes toward a Sept. 20 kickoff for rolling out the extra shots. The panel will continue to follow the usual processes in its consideration of boosters, with a vote on recommendation in the “very near future,” said Grace Lee, the panel’s chairperson. Read more from Fiona Rutherford.
Biden Urges States Pay Low-Income People to Get Shots: The Biden administration is encouraging states to offer gift cards or similar incentives to low-income people on Medicaid who get the Covid-19 vaccine, in a bid to boost inoculation rates that trail the broader public. The Centers for Medicare & Medicaid Services issued new guidance yesterday to state Medicaid directors, offering federal funding and other assistance to boost vaccination rates among people covered by the health plan, Josh Wingrove reports.
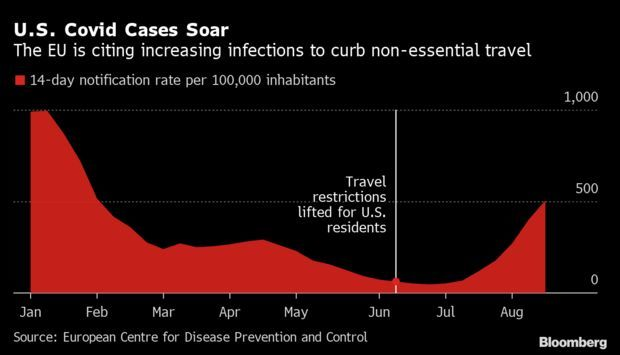
EU Reimposing Travel Curbs on Americans: European Union countries voted to subject the U.S. to fresh restrictions on travel as America faces a surge in Covid-19 cases, dealing another blow to the tourism industry. A qualified majority of ambassadors voted to reintroduce the curbs, which were lifted in June, the EU said yesterday. The limits will primarily impact unvaccinated Americans. The U.S.’s 588 new cases per 100,000 this month is far above the EU’s limit of 75. Jasmina Kuzmanovic and Andra Timu have more.
More Headlines:
- Moderna Falls as Japan Probes Two Deaths, FDA Flags Heart Risk
- China Misses Target for BioNTech-Pfizer Shot, Raising Questions
- Need PCR Tests for Your Next Luxury Trip? That Could Be $6,000
- NRx’s Zyesami Indicates Improved Lung Function With Covid-19
What Else to Know Today
More Americans Covered by Health Insurance in 2020, CDC Says: U.S. health-insurance coverage improved last year, with more than 90% of Americans on a plan, the Centers for Disease Control and Prevention said. The number of uninsured fell to 31.6 million, or 9.7% of the population, in 2020, according to preliminary estimates released by the CDC’s National Center of Health Statistics Tuesday. In 2019, 33.2 million, or 10.3%, were uninsured. Read more from Alex Tanzi.
Abortion Clinics Ask SCOTUS to Halt Near-Total Texas Ban: Abortion providers asked the U.S. Supreme Court to halt a Texas law that’s set to take effect tomorrow and would outlaw the procedure as early as six weeks into pregnancy. The emergency filing sets up what could be a telling showdown at a conservative-controlled bench that is already planning to hear a Mississippi appeal that seeks to reverse the core constitutional right to abortion. The Texas law would be the strictest in the nation, barring abortion after a fetal heartbeat can be detected—and before many women know they are pregnant. The challengers say the law would ban abortion for at least 85% of patients in the state. Greg Stohr has more.
Energy Panel Republicans Worried About Reconciliation: House Energy and Commerce Committee Republicans don’t want the panel’s markup process for reconciliation rushed, calling Sept. 15 an arbitrary deadline. The committee’s instruction per the resolution is $486.5 billion as part of Democrats’ $3.5 trillion tax and spending plan. The members wrote in a letter to Chair Frank Pallone (D-N.J.) that “any markup of legislation to meet the Committee’s Budget Reconciliation instructions must occur when Members are present in Washington, DC and can meet in person to conduct the markup. During the last Reconciliation markup, the Committee operated virtually and just before a holiday weekend. Given the arbitrary deadline of September 15th is fast approaching, we are concerned that Democrats plan to repeat this sham process.”
Purdue Pharma Tests Limits of Liability Shields: Purdue Pharma’s insistence on a bankruptcy settlement that would protect its billionaire owners from civil lawsuits related to OxyContin’s role in the opioid epidemic—while allowing the Sacklers to avoid filing for bankruptcy themselves—is pushing the limits of a common tool in Chapter 11 cases. Purdue’s owners said they will contribute $4.35 billion to the company’s bankruptcy estate only if the family and hundreds of others also get immunity. Daniel Gill has more.
New HHS Office Targets Climate, Health Equity: The Health and Human Services Department announced a new office to help safeguard the health of people bearing the brunt of climate change, a day after Hurricane Ida hit the Gulf Coast. The first-of-its-kind “Office of Climate Change and Health Equity” will focus on protecting the health of those “experiencing a disproportionate share of climate impacts and health inequities,” Secretary Xavier Becerra said yesterday. Read more from Shira Stein.
ACA’s Author Returns to HHS: Dara Corrigan, who oversaw the crafting of the regulations that overhauled the private insurance market, has joined the U.S. Centers for Medicare & Medicaid Services as deputy administrator and head of the Center for Program Integrity, the agency told Bloomberg Law. Corrigan, who also served as associate commissioner for global regulatory policy at the FDA, will be tasked with protecting federal Medicare and Medicaid programs from fraud, waste, and abuse. Shira Stein has more.
Sutter to Pay $60 Million More to Settle Medicare Billing Claims: California health-care giant Sutter Health and several affiliates will pay an additional $60 million to settle claims they knowingly inflated how sick Medicare beneficiaries were to boost reimbursements, federal prosecutors announced yesterday. Read more from Joyce E. Cutler.
More Headlines:
- HHS Secretary Backs EPA’s February Ban on Chlorpyrifos in Food
- Perspecta, Capgemini Nab $2 Billion Defense Health IT Contract
- Poseida Says FDA Cleared IND Application for Cancer Treatment
- Recreational Pot Backers Permitted to Collect Signatures in Ohio
- FDA Provides Lantern Addl Orphan Drug Designation for LP-184
To contact the reporter on this story: Brandon Lee in Washington at blee@bgov.com
To contact the editors responsible for this story: Zachary Sherwood at zsherwood@bgov.com; Giuseppe Macri at gmacri@bgov.com; Michaela Ross at mross@bgov.com
Stay informed with more news like this – from the largest team of reporters on Capitol Hill – subscribe to Bloomberg Government today. Learn more.