HEALTH CARE BRIEFING: States See More Hospitalized Young People
Bloomberg Government subscribers get the stories like this first. Act now and gain unlimited access to everything you need to know. Learn more.
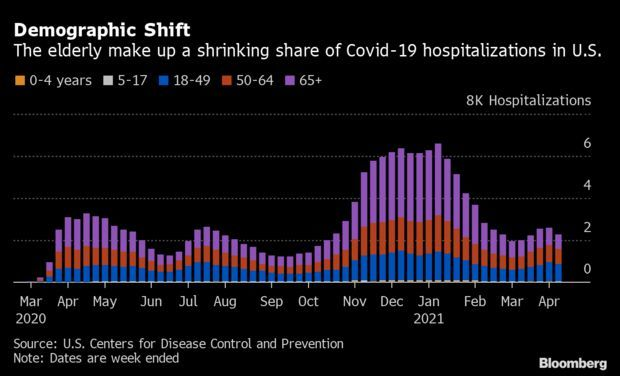
Throughout previous waves of Covid-19, children and young adults eluded the coronavirus’s gravest consequences, exhibiting mild symptoms or none at all. Now faster-spreading strains are sending more to the hospital. Fatalities remain low, but doctors say the virus is now making young people sicker, including some gravely.
In Michigan, where Covid-19 is spreading at one of the planet’s fastest rates—an average of almost 8,000 new cases a day—hospitals see far more youths. “Statewide, the hospitalization rates are higher, therefore they must be sicker,” said Rudolph Valentini, chief medical officer for Children’s Hospital of Michigan in Detroit. “We are seeing more kids coming to ER across the state. Some are in ICU, and some require mechanical ventilation.”
The new Covid-19 variants are more contagious and hit young people harder than the original virus that emerged more than a year ago, when people under 30 often showed no symptoms. In the 5- to 17-year-old age group, there have been only 293 recorded deaths from Covid-19 in the U.S., or about 0.1% of the country’s overall deaths, according to data from the Centers for Disease Control and Prevention.
Now, however, “We are seeing patients 20, 25, 35 who are quite ill with Covid,” Robert Riney, chief operating officer for Henry Ford Health System, said in a briefing last week. “The good news is the mortality rates are lower, but these are not light cases.”
And the same trend is showing up across the country as young people, who often aren’t yet vaccinated, spread and contract the virus through school sports and social gatherings. Most states have focused vaccination efforts on residents over 65 because they’re most vulnerable.
The next problem might be cases of Multisystem Inflammatory Syndrome, in which multiple organs become inflamed. MIS-C, as it’s called, afflicts some Covid-19 survivors with lingering symptoms such as higher heart rates, body aches and fatigue. Though the syndrome is rare, hospitals likely will see more of it after the variant-driven surge, because it trails Covid-19 by two to five weeks, said Valentini. Read more from David Welch, Sarah Kopit and Nic Querolo.
Happening on the Hill
House Panel Launches Emergent Probe on Vaccine Deals: The House Oversight and Reform Committee and the Select Subcommittee on the Coronavirus Crisis launched an an investigation into Emergent BioSolutions’ performance under federal vaccine contracts, according to a statement from panel Chairs Carolyn Maloney (D-N.Y.) and Jim Clyburn (D-S.C.). The lawmakers are probing whether the company leveraged its relationship with a key Trump Administration official to profit from federal contracts despite a track record of raising prices and failing to meet contract requirements, and whether its actions impeded the pandemic response. A hearing on the matter is slated for May 19, Max Zimmerman reports.
The chairs also said they were “investigating Emergent’s actions to unduly influence anthrax vaccine assets currently stockpiled in the Strategic National Stockpile,” according to a statement.
- Meanwhile, Emergent BioSolutions was told by U.S. regulators to stop making Johnson & Johnson’s Covid-19 vaccine at a facility where 15 million doses worth of a key ingredient had to be discarded, adding to roadblocks preventing wider use the single-dose shot. Use of J&J’s vaccine has been paused since last week as health officials probe a possible link to life-threatening blood clots. The halt at Emergent, a contract manufacturer, could add to questions about how many Americans will ultimately receive J&J’s shot even if it is found to be safe. Read more from Riley Griffin.
House Passes Marijuana Banking Bill: The House passed a bill that would give state-authorized marijuana businesses easier access to banking services. The bill (H.R. 1996), called the SAFE Banking Act, would prohibit federal banking regulators from penalizing banks and other depository institutions for providing banking services to cannabis businesses. It passed last night on a 321-101 bipartisan vote. Read more from Jarrell Dillard and Tiffany Kary. Read the BGOV Bill Summary by Michael Smallberg.
Covid-19 Recovery: The Senate Health, Education, Labor and Pensions Committee holds a hearing today on recovering from Covid-19 through workforce modernization training, education and employment.
Democrats Prod Facebook, Twitter on Vaccine Info: Sens. Amy Klobuchar (D-Minn.) and Ben Ray Luján (D-N.M.) in a Friday letter to Facebook CEO Mark Zuckerberg and Twitter CEO Jack Dorsey asked that the companies remove accounts belonging to 12 users said to be responsible for 65% of anti-vaccine disinformation content on the platforms — findings the lawmakers attributed to a new report from the Center for Countering Digital Hate.
“For too long, social media platforms have failed to adequately protect Americans by not taking sufficient action to prevent the spread of vaccine disinformation online,” the senators wrote, according to a statement. “An earlier report found that during the coronavirus pandemic, social media platforms failed to act on 95 percent of such disinformation reported to them.”
The Coronavirus Pandemic
Vaccinations Study Highlights How Shots Curb Spread: Covid-19 infection and death rates in Israel, the world’s most vaccinated major country, are falling roughly in line with the order in which various age groups gained eligibility to get their shots, according to a study showing the campaign’s impacts. While all Israelis endured a nationwide lockdown and rules on social distancing, the age-tiered declines seen in the study suggest vaccines are playing a major role in blunting the impact of the pandemic.
Around 55% of Israelis are now vaccinated, giving it a unique standing among researchers as governments globally seek signs their vaccine campaigns are working. In the U.S., with about 32% of people fully immunized, early data suggests that similar trends may be seen as states expand vaccine eligibility to those as young as 16, meeting the Biden administration deadline of April 19. Read more from John Lauerman.
NIH to Fund Trial on Drugs Repurposed for Covid-19: The National Institutes of Health will provide $155 million in funds for a Phase 3 clinical trial that tests several existing prescription and over-the-counter drugs for people to self-administer to treat symptoms of Covid-19. The trial aims to provide treatment options to the majority of adult Covid-19 patients with mild-to-moderate cases who are not sick enough to be hospitalized. Read more from Sebastian Tong.
More U.S. Headlines:
- State Dept. to Caution Against Travel for About 80% of Countries
- United Betting on Three Summer European Spots for Americans
- HHS Should Clarify Roles for Citizens’ Emergency Return During a Pandemic (GAO)
More Global Headlines:
- Pfizer Backed Down From Demands Called ‘Risk’ to South Africa
- Chile Reaches Deals for Third Doses of Sinovac, Pfizer If Needed
- Philippines Clears Coronavirus Vaccines From J&J, India’s Bharat
- China Suspicion, ‘Foreign Plot’ Fears Hamper Africa Vaccine Plan
What Else to Know
U.S. May Curb Nicotine, Rattling Cigarette Makers: The Biden administration is mulling a requirement that nicotine be lowered to levels that are not addictive, The Wall Street Journal reports, citing people familiar with the matter. The Food and Drug Administration could also move to ban menthol cigarettes, it reports. Tobacco producer Altria said that any FDA action “must be based on science and evidence and must consider the real-world consequences of such actions, including the growth of an illicit market.” The company said it will “continue to engage in this long-term regulatory process.”
The FDA has been expected to state its position on whether it’ll still permit menthol cigarettes by April 29, under a deadline imposed by a lawsuit. The case, brought by the African American Tobacco Control Leadership Council, had sued the government agency saying it had taken an unreasonably long time to respond to a citizen’s petition filed several years ago that sought to prohibit menthol as a flavor in cigarettes. Read more from Tiffany Kay.
Federal and State Health Waiver Talks Less Indulgent: The Biden administration’s rapid actions to undo state work requirements for Medicaid breaks from the generally indulgent past approach to pilot health programs, signaling upcoming transformations in the waiver system. The Health and Human Services Department’s recent notices to several states that HHS is rolling back Trump-era must-work rules for adults on Medicaid is straight out of the administrative-law playbook, involving public notice and opportunities for review hearings. Read more from Christopher Brown.
J&J, Teva Opioid Trial May Signal Cost of Settlements: A California judge may soon provide a clearer picture of what it may cost drug corporations including Johnson & Johnson and Teva Pharmaceutical Industries to resolve liability for their role in the nationwide opioids epidemic. The first trial against the opioid companies in nearly two years began yesterday in Santa Ana, where four California municipalities are demanding at least $50 billion for what they said was the illegal marketing of pain pills. Read more from Jef Feeley.
Butterfly Hires Former Ernst Staffer: Digital health company Butterfly Network hired Andrea Hechavarria to serve as its vice president of government affairs, leading its lobbying in Washington. She most recently worked within the lobbying shop at 3M and, before that, served as a staffer to Sen. Joni Ernst (R-Iowa), and former Sens. Mike Johanns (R-Iowa), Judd Gregg (R-N.H.) and John Sununu (R-N.H.), Megan R. Wilson reports.
More Headlines:
- Trans Teens in Arizona Appeal Decision Blocking Medicaid Pay
- Galectin Rises After Hiring Ex-HUD Secretary Carson to Consult
- Amgen Gastric Cancer Drug Gets Breakthrough Therapy Label
- Alnylam Submits New Drug Application on Vutrisiran With FDA
With assistance from Megan R. Wilson
To contact the reporter on this story: Brandon Lee in Washington at blee@bgov.com
To contact the editors responsible for this story: Zachary Sherwood at zsherwood@bgov.com; Giuseppe Macri at gmacri@bgov.com; Michaela Ross at mross@bgov.com
Stay informed with more news like this – from the largest team of reporters on Capitol Hill – subscribe to Bloomberg Government today. Learn more.