HEALTH CARE BRIEFING: Moderates Eye Vaccine Bill Before Stimulus
Bloomberg Government subscribers get the stories like this first. Act now and gain unlimited access to everything you need to know. Learn more.
Speaker Nancy Pelosi (D-Calif.) is under pressure from moderate Democrats, whose votes will be needed to pass any stimulus bill along party lines, to split up a vaccination funding package from President Joe Biden’s broader Covid-19 relief bill.
The Blue Dog Coalition of Democrats wrote in a letter to Pelosi published yesterday that the current plan to pass the administration’s $1.9 trillion stimulus bill through a special budget process could take “months.” But funding for the patchy U.S. vaccination effort is necessary right away, they argued.
“Vaccine distribution is a hot mess left by the Trump administration,” Rep. Stephanie Murphy (D-Fla.), one of the Blue Dog Democrats co-chairs, said in an interview. “Fixing it now is something the Biden administration and Democrats can do quickly to show we’re delivering for the American people.”
In the closely divided House, Pelosi can afford to lose no more than four votes on a party-line vote if all members vote. Pelosi’s office didn’t immediately respond to a request for comment on the letter, which was signed by Murphy and fellow co-chairs Ed Case (D-Hawaii), Kurt Schrader (D-Ore.) and Tom O’Halleran (D-Ariz.).
“Our preferred legislative approach is to swiftly vote on the portion of the administration’s proposal focused on vaccine development and distribution,” the group wrote in their letter. “Other larger, complementary elements” of the president’s relief could move as quickly as possible on a separate track, they wrote.
The Blue Dogs argue that vaccine funds can easily pass Congress even as work continues on the stimulus measure. Murphy said that if it couldn’t pass by unanimous consent, the House should change its schedule to vote on it as quickly as possible. The chamber is slated to leave town today, staying out until Feb. 23. Read more from Erik Wasson.
Also Happening on the Hill
Senate Adopts Blueprint for Major Stimulus as Harris Breaks Tie: The Senate voted 51-50, after Vice President Kamala Harris broke her first tie, to adopt a budget blueprint for Biden’s $1.9 trillion virus relief package — following nearly 15 hours of wading through amendments from both parties.
The House had already adopted its budget resolution but will likely have to vote again today to agree on the Senate’s language. Once that’s done, Democrats will be able to craft a relief bill in the coming weeks that can pass without any Republican votes under special budget rules — though the White House, moderates like Sen. Joe Manchin (D-W.Va.) and others still say they want a bipartisan final product.
House and Senate committees would have until Feb. 16 to write the stimulus legislation under the instructions in the budget. The final action followed a marathon of votes on amendments known as a vote-a-rama. Most of the non-binding measures were intended more to make points on hot-button issues like taxes, abortion, immigration and schools that had little or sometimes nothing to do with pandemic aid. Read more from Steven T. Dennis.
Covid-19 Testing Mandate Idea for Domestic Flight Debated: Lawmakers in both parties and representatives from the aviation industry say mandated Covid-19 testing for domestic flights would lead to even greater furloughs and losses, especially if federal aid to keep airline employees on the payroll expires next month. The bipartisan opposition comes a week after Marty Cetron, director of the Centers for Disease Control and Prevention’s Division of Global Migration and Quarantine, said at a briefing that the agency was “actively looking” into a Covid-19 testing requirement for domestic flights. Read more from Lillianna Byington.
Organ Donation Network Draws Scrutiny: A bipartisan Senate duo subpoenaed the United Network for Organ Sharing for documents concerning oversight of organ transplant centers. Sens. Chuck Grassley (R-Iowa) and Ron Wyden (D-Ore.) wrote they “have serious concerns” about the private nonprofit group’s oversight of organizations responsible for procuring organs, which they argued “have been severely underperforming for decades.” Read more from Peyton Forte.
Warner, Warnock Introduce Medicaid Expansion Bill: Eleven Senate Democrats led by Sens. Mark Warner (D-Va.) and Raphael Warnock (D-Ga.) introduced a measure yesterday to encourage states to expand their Medicaid program. The bill would permit states that bolstered Medicaid after 2014 or ones that plan to “expand Medicaid in the years ahead” to receive the same full federal match as the states that expanded Medicaid earlier under the terms of the Affordable Care Act. Read the legislation here.
Orphan Drug Loophole Gets Senate Scrutiny: A bipartisan group of senators, Bill Cassidy (R-La.), Tammy Baldwin (D-Wis.) and Jeanne Shaheen (D-N.H.), is seeking to “close the orphan drug loophole that drug companies use to keep competition off the market and rake in profits.” The three lawmakers unveiled a bill yesterday that aims to let the FDA rescind exclusivity given to drugmakers if they prove a drug isn’t economically viable. Read the legislation here.
N.J. Lawmakers Call for Nursing Home Oversight: Reps. Bill Pascrell (D-N.J.) and Mikie Sherrill (D-N.J.) and Sen. Bob Menendez (D-N.J.) yesterday unveiled legislation to restore regulations “regarding infection control and emergency preparedness” in nursing home facilities. The bill would require such facilities to “establish and maintain an infection prevention program and an emergency plan to protect residents” in an emergency situation. Read the legislation here.
Report Says Baby Foods Test Too High for Toxic Metals: A new congressional report found that commercial baby foods are often tainted with significant amounts of heavy metals, from arsenic to lead to mercury, prompting lawmakers to recommend more stringent testing and labeling, among other suggestions. The study, released by the House Oversight Committee’s Consumer Policy Subcommittee, recommends the FDA set a maximum of toxic heavy metals permitted in baby foods. Read more from Anne Riley Moffat.
The Coronavirus Pandemic
Johnson & Johnson Submits FDA Application: Johnson & Johnson asked U.S. drug regulators to clear its experimental Covid-19 vaccine for emergency use, setting up what is poised to be a fast-moving review process that could lead to millions more doses becoming available to step up a stumbling immunization effort. The drugmaker in a statement said it filed an application for an emergency-use authorization with the Food and Drug Administration. If cleared, the J&J shot would offer the U.S. a third vaccine to try to halt a disease that’s killed more than 450,000 Americans.
The FDA scheduled a Feb. 26 meeting at which a panel of outside advisers will consider the request for clearance and make a recommendation about whether to grant it. While its decision isn’t binding, it is unusual for the FDA to reject the experts’ advice. Read more from Riley Griffin.
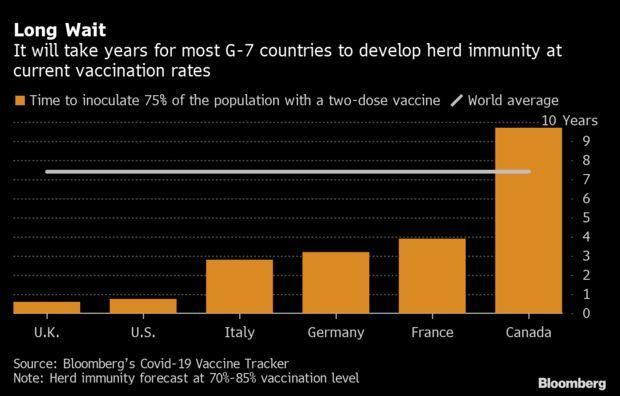
U.S., Israel Years Ahead of Europe in Immunity Effort: The U.S. is on pace to vaccinate 75% of its population against Covid-19 this year, while Canada will need almost a decade to reach that coverage level, according to the Bloomberg Vaccine Tracker. Much of Europe would also need several years for herd immunity, whereas the U.K. and Israel are on track to achieve the 75% mark this year. Read more from Tim Loh.
Vaccines From Nursing Homes Boost Supply in States: Dozens of U.S. states are releasing thousands of vaccines left over from a federal initiative for nursing homes, increasing supply for other eligible people including essential workers and seniors. Thirty-two states and cities are transferring their extra doses from the federal program, a spokesperson for the Centers for Disease Control and Prevention said. Angelica LaVito has more.
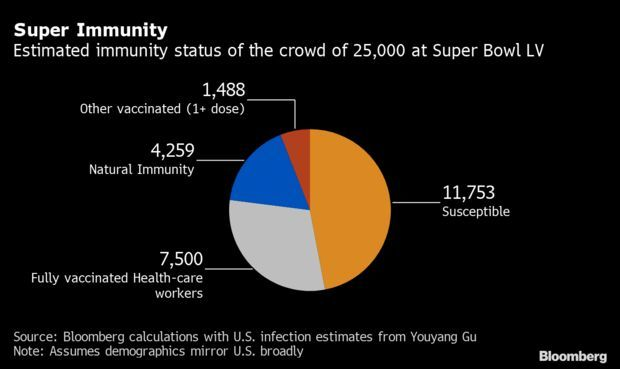
Super Bowl Crowd Presents Immunity Experiment: The National Football League is hoping to avoid any large-scale Covid-19 outbreaks at the Super Bowl, and immunity appears to be a major part of the game plan. A sharp drop in new infections also is on the league’s side. The NFL capped overall attendance at 25,000, a record low for football’s biggest event, which this year features the Kansas City Chiefs and Tampa Bay Buccaneers in Tampa, Florida.
While the league has given tickets to 7,500 vaccinated health-care workers for the big game, it hasn’t released health information about the other attendees. But statistical models can help gauge the risk. Among the attendees who got tickets on their own, 8.5% have likely benefited from at least one of the required two doses of vaccine, and about 27% would likely have been previously infected, based on Bloomberg Vaccine Tracker data and cumulative estimates from an independent modeler. Jonathan Levin and Michael Smith have more.
More U.S. Headlines:
- Coronavirus Testing Experiment in Schools Hits Logistical Snarls
- FDA, Citing Safety, Warns of Compounding Remdesivir Products
- Coronavirus Death Claim Shut Out of Federal Court in California
- Wisconsin Governor Strikes Back at GOP Move on Face-Mask Mandate
Global Headlines:
- Merkel Warns of Easing Lockdown in ‘Final Stretch’ of Pandemic
- Sweden Sees Rollout of Digital Vaccine Certificates This Summer
- Macron Asks China for More Transparency on Covid-19 Vaccines
What Else to Know Today
Drug Industry Group on Hunt for Democratic Lobbyist: The Pharmaceutical Research and Manufacturers of America is on the search for a new lobbyist specializing in relationships with Senate Democrats, according to a listing on LinkedIn posted yesterday. The person’s portfolio should include speaking with lawmakers and staff about “potential/proposed legislation and the impact of such legislation to patients and the pharmaceutical industry,” it says.
The post comes a month after Democrats took back control of the Senate, following a pair of runoff elections in Georgia. In the final months of 2020, PhRMA also lost a House Democratic lobbyist. Erin Mendelsohn, a former aide to Rep. Anna Eshoo (D-Calif.) who served as senior director of federal advocacy at PhRMA, became director of federal affairs at Takeda Pharmaceutical Company, Megan R. Wilson reports.
Opioid Focus Will Be on Treatment, Drug Czar Says: The Biden administration will focus on recovery treatment programs in tackling the opioid crisis that has surged amid the pandemic, setting the tone for the White House as it embarks on the latest chapter of the battle against addiction. The new focus signals a change from Trump’s White House, which had been more skeptical of harm-reduction approaches to treatment, such as needle exchanges. Read more from Ian Lopez and Valerie Bauman.
Former HHS Official Sues Biden to Keep Post: Roger Severino was appointed to an advisory committee just days before Trump left office. Now, he’s suing the new president to hold onto his job. The former director of the Health and Human Services Department’s civil rights office filed a lawsuit on Wednesday after receiving an email from the new administration that asked him to step down from the Council of the Administrative Conference. Lydia Wheeler has more.
More Headlines:
With assistance from Megan R. Wilson
To contact the reporter on this story: Brandon Lee in Washington at blee@bgov.com
To contact the editors responsible for this story: Zachary Sherwood at zsherwood@bgov.com; Giuseppe Macri at gmacri@bgov.com; Michaela Ross at mross@bgov.com
Stay informed with more news like this – from the largest team of reporters on Capitol Hill – subscribe to Bloomberg Government today. Learn more.