HEALTH CARE BRIEFING: Democrats Weigh Voiding Trump Health Rules
Bloomberg Government subscribers get the stories like this first. Act now and gain unlimited access to everything you need to know. Learn more.
Democrats are considering which Health and Human Services Department regulations they may want to overturn in the early days of the Biden administration, although there are few candidates and other avenues to do so.
Their main candidates for congressional reversal are a drug rebate rule that removes legal shields for reimbursements paid by drug companies to middlemen and insurers and a rule requiring the department review all previous regulations or otherwise the rules must expire.
The Congressional Review Act lets lawmakers nullify federal regulations shortly after they’re enacted. Its use has been floated amid a flurry of last-minute rulemaking from the outgoing Trump administration, particularly from HHS. It would only require a simple majority in each chamber to pass a resolution disapproving of a rule and effectively rescinding it.
But many of the Trump administration health regulations Democrats would want to reverse are outside the window of time where they can do so. And with limited floor time, they may target other policymaking areas. This is the one of the shortest windows of time to use the CRA due to Congress being in session for so many days at the end of the year, said Matthew Kent, a regulatory policy associate with consumer advocacy group Public Citizen.
Congress will likely be able to look at rules dating back to the third week of August, said Kent. “I think of the CRA as a legislative ‘In Case of Emergency, Break Glass’ option for legislators,” he said.
The Biden administration may also reopen public comment, do additional rulemaking, or decide not to enforce the regulations.
“Congress doesn’t use the CRA randomly” and is calculated about doing so, according to Mike Strazzella, head of the federal government relations practice at Buchanan, Ingersoll & Rooney. Read more from Shira Stein and Alex Ruoff.
The Coronavirus Pandemic
Trump Administration Plans to Speed Up Vaccinations: Trump administration officials today plan to strongly encourage states to expand access to Covid-19 vaccinations to anyone older than 65, regardless of underlying conditions, and any American 16 and older with a pre-existing condition, according to two U.S. officials. Dr. Robert Redfield, director of the Centers for Disease Control and Prevention, will send out a letter to governors making these recommendations today. Existing guidelines from the Advisory Committee on Immunization Practices won’t be changed, one official said. Axios first reported that an announcement would be coming. Read more from Jennifer Jacobs.
Meanwhile, the tally of Covid vaccine doses administered in the U.S. rose by 1.25 million yesterday, a record daily gain amid what’s been an otherwise slow rollout of the shots. Bloomberg is counting vaccine doses administered in the U.S. and around the world, collecting data from state vaccine reports and the Centers for Disease Control and Prevention. Because the CDC and some states don’t report on the weekend, Monday’s tally includes catch-up from prior days. Drew Armstrong has the latest on the figures.
Vaccine Skepticism Extends Even to Health Workers: In North Carolina and Ohio, nursing-home employees are declining the Covid-19 vaccine. A Florida physician and a New York City paramedic are saying no. Federal agencies, which left the states to sort out immunizations, is suggesting pizza parties and raffle prizes as incentives for medical staffers to roll up sleeves. Reluctance to get the shot that could end the pandemic goes well beyond “anti-vax” activists who spew baseless theories online. Nurses and firefighters are among those questioning its safety after approval in record time. Read more from Elise Young.
- Also hampering vaccine efforts are signup processes that are confusing to the elderly. New York City residents hoping to get vaccines are confronting a “bewildering signup process” that is slowing plans to hasten inoculations, the city’s comptroller said. New York launched mass vaccination sites yesterday in Brooklyn and the Bronx, expanding shots yesterday to people 75 and over. Read more from Stacie Sherman.
Pfizer Boosts Vaccine Goal to 2 Billion Doses: Pfizer and BioNTech raised their coronavirus vaccine production target for this year to 2 billion doses, as the partners respond to a global rush of countries seeking to order more doses. The drug companies have already committed more than half that capacity, BioNTech announced. A more than 50% hike from the previous 2021 target of 1.3 billion doses, the increase incorporates a label change that’ll allow doctors to extract six doses, rather than five, from every vial, BioNTech said. Naomi Kresge has more.
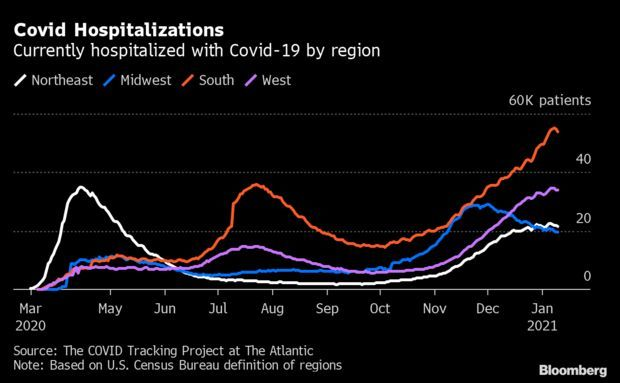
Northeast Sees Signs of Easing Hospitalizations: The pace of Covid-19 hospitalizations in the Northeast is showing some preliminary signs of easing, adding to hopeful indicators in the Midwest, where the latest viral wave started. In the Northeastern states, the number of patients currently hospitalized with Covid-19 was 21,494 as of Sunday, up 0.8% from a week earlier, the smallest one-week percentage increase since September, according to the Covid Tracking Project. Read more from Jonathan Levin.
Democrat to Hospital with Virus, Blames Maskless Colleagues: Rep. Bonnie Watson Coleman (D-N.J.), a 75-year-old lung-cancer survivor, is seeking hospital treatment after testing positive for Covid-19. Watson Coleman believes she was exposed after sheltering with several maskless colleagues during last week’s riot at the Capitol, according to a statement. She said yesterday she received a positive rapid test and is awaiting the results of PCR testing. Video of the attack shows multiple Republican lawmakers refusing masks as they were ushered into an enclosed area. Sunday, the Capitol’s attending physician warned that lawmakers may have been exposed to an occupant in the room who was infected with the coronavirus. Elise Young has more.
Meanwhile, Rep. Pramila Jayapal (D-Wash.) said she also has tested positive for Covid-19, days after having shared a safe room with Republican colleagues who declined to wear masks. “I just received a positive COVID-19 test result after being locked down in a secured room at the Capitol where several Republicans not only cruelly refused to wear a mask but recklessly mocked colleagues and staff who offered them one,” she wrote in Twitter post, Derek Wallbank and Dina Bass report.
Jayapal said she’s calling for “serious fines to be immediately levied on every single Member who refuses to wear a mask in the Capitol” and for lawmakers refusing to wear a mask to be “immediately removed from the floor by the Sergeant at Arms.”
More Headlines:
- Biden Gets Second Shot and Faults Mask-Rejecting Republicans
- HHS Seeks to End Oversight of Some Gloves, Masks, Ventilators
- Deliveries of Vaccines to EU Will Begin on Jan. 11, Moderna Says
- Moderna Says It’s Expanding Vaccine Pipeline With Three Plans
- Quanterix Protein Antigen Test for Virus Gets FDA Authorization
What Else to Know
Separated Migrant Mental Health Help Delayed by DHS, Lawyers Say: Lawyers for migrants separated from their families at the border say the Trump administration has delayed efforts to provide them mental health services, potentially causing them more harm. A contractor hired by the government in March has reached just 561 of the 2,103 parents and children identified by a California judge as deserving mental health services after being separated from each other upon entering the U.S., according to government documents released this month.
That figure would be much higher if the Department of Homeland Security hadn’t held back contact information for eligible migrants or if the government had settled the case earlier, lawyers for the families say. Read more from Alex Ruoff.
FDA Chief Counsel Steps Down: The FDA’s senior attorney, Stacy Cline Amin, stepped down yesterday, the agency’s head announced. Amin has served as chief counsel for the Food and Drug Administration since 2018, coming to the agency after a year as chief associate counsel in the Trump administration and after counsel positions in both the House and Senate. Mark Raza, who has been the principal deputy chief counsel since 2011, will take over as the acting counsel. Read more from Jeannie Baumann.
More Headlines:
- Fetal Tissue Studies Need Donor Consent Under HHS Proposal
- U.S. Will Pump $8 Million Into Broadband for Telehealth Access
- Eli Lilly Surges as Experimental Alzheimer Drug Shows Promise
- Crest Toothpaste Misleadingly Says It’ll Repair Gums, Suit Says
- Zantac Brand Name Makers’ Design Defect Claims Thrown Out
To contact the reporter on this story: Brandon Lee in Washington at blee@bgov.com
To contact the editors responsible for this story: Zachary Sherwood at zsherwood@bgov.com; Giuseppe Macri at gmacri@bgov.com; Michaela Ross at mross@bgov.com
Stay informed with more news like this – from the largest team of reporters on Capitol Hill – subscribe to Bloomberg Government today. Learn more.