HEALTH CARE BRIEFING: Democrats Look to Biden on Vaccine Funds
Bloomberg Government subscribers get the stories like this first. Act now and gain unlimited access to everything you need to know. Learn more.
Democrats in Congress are looking to President-elect Joe Biden for more spending in 2021 to address the coronavirus pandemic and boost the economy.
Vaccine distribution funding is among the leading priorities for Democrats, with control of the Senate, which hinges on tomorrow’s Senate runoffs in Georgia, set to be a key factor in some of those debates.
Current coronavirus vaccine distribution funding isn’t enough, said Jen Psaki, Biden’s pick for press secretary, last week. Biden wants more mobile vaccination sites and a bigger outreach campaign to raise awareness, she said. “That is why the president-elect is going to be coming back and talking about a new package and what is needed in the new year,” Psaki said.
“This has to be done better,” Speaker Nancy Pelosi (D-Calif.) said last week about the vaccine distribution efforts. She said she backs Biden’s goal for “100 million vaccinations in a short period of time,” including the use of the Defense Production Act.
Congress provided $8.75 billion for the Centers for Disease Control and Prevention for vaccine distribution in its December coronavirus aid package—of which $4.5 billion would be for states and local governments, as well as territories and tribes, Alex Ruoff and Jack Fitzpatrick report.
- Read more: Biden Faults Trump for Slow Vaccine Rollout, Vows Faster Pace
- Read more: Sluggish Vaccine Campaign Raises Specter of U.S. Dysfunction
Sen. Mitt Romney (R-Utah) on Friday also criticized the slow rollout of the vaccines in the U.S., blaming the federal government for not helping U.S. states to administer them. “That comprehensive vaccination plans have not been developed at the federal level and sent to the states as models is as incomprehensible as it is inexcusable,” Romney said in a statement.
About 3.2 million doses have been administered, far short of the Trump administration’s goal of administering 20 million vaccinations by the end of 2020. Officials have pinned delays on a delicate vaccine with complex storage requirements, uncertainty over the supply of doses and strain on local health agencies already facing historic challenges. David McLaughlin has more.
Still, the U.S. government’s top infectious diseases doctor said yesterday the vaccine rollout is picking up speed and could be fully on track in a week or so. “It’s just trying to get a massive vaccine program started and getting off on the right foot. The important thing is to see what is happening in the next week, to week and a half,” Anthony Fauci told ABC. Read more from Todd Shields and Yueqi Yang.
Happening on the Hill
New Congress Begins: The 117th Congress began yesterday and business is expected to be light through this week, as lawmakers reorganize committees and adopt new rules for floor proceedings. Congress will convene to certify Electoral College results on Wednesday. Watch for hearing announcements from relevant committees as they prepare to consider Biden’s Cabinet nominees.
House Changes Budget Requirements for Virus Bills: The House will vote today on the rules package (H. Res. ?) governing proceedings for the 117th Congress, which would exempt legislation dealing with the Covid-19 pandemic and climate change from budget rules including pay-as-you-go or PAYGO requirements. PAYGO typically requires provisions that would increase the deficit to be offset with spending cuts or revenue increases elsewhere. The House re-adopts its rules every two years at the beginning of a Congress and the majority takes the opportunity to make changes to the chamber’s operations.
Progressives had urged Democratic leaders to change PAYGO as part of the package. The resolution wouldn’t amend the rule, though it would allow the House Budget Committee chairman to exclude the budgetary effects of qualifying coronavirus and climate provisions from estimates of the legislation’s impacts on budget authority, outlays, mandatory spending, entitlement authority, and federal revenue. Read more from Loren Duggan.
The Coronavirus Pandemic
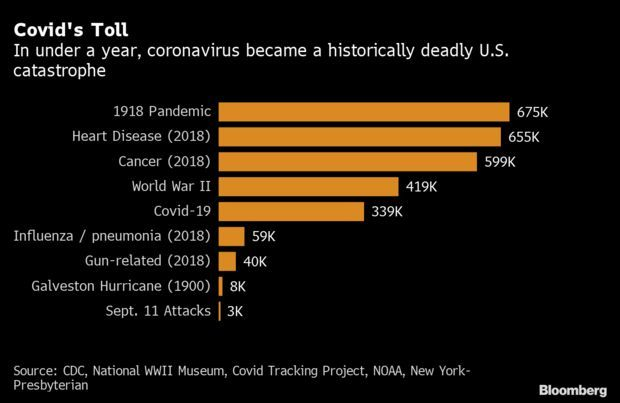
Covid-19 Joins War, Cancer as Historic Blight: As 2020 came to a close, most will remember the year as one defined by sacrifice and loss. In the 347 days since the first reported U.S. case, the novel coronavirus officially called SARS-CoV-2 bloomed into one of the textbook disasters of modern history. By casualty count, the country is mourning the equivalent of the death toll of the Sept. 11 attacks each day, and is expected to for months. In totality, 1 out of every 1,000 Americans have died, with heavier burdens on seniors and minorities. Nic Querolo has more.
Distilleries Making Sanitizer Get Fee Relief: U.S. distilleries and other groups that began to make hand sanitizer as the pandemic gripped the country have gotten relief from thousands of dollars in user fees normally levied against companies that produce medical products. The craft beverage industry, largely made up of small businesses, got unwelcome news last week in the Federal Register after the FDA indicated it may charge up to $14,000 to companies that made hand sanitizer. Read more from Michelle Fay Cortez.
More Coronavirus Headlines:
- Underused Covid-19 Therapy Gets Boost in Nursing Homes From HHS
- A Trump-Touted Covid-19 Therapy Awaits Proof to Back Up His Boasts
- Pipette Tips Needed for Covid-19 Testing in Short Supply, FDA Says
- Biden’s Inaugural Ceremonies to Include Memorial to Virus Victims
- Why the U.K.’s Mutated Coronavirus is Fanning Concerns: QuickTake
More on Vaccines:
- Vaccine Public Confidence Media Blitz Coming from HHS in January
- Emergency Virus Vaccine Approval Adds More Risk to Job Mandates
- Religious Objections to Vaccines Will Clash With Employer Defense
What Else to Know
Surprise Billing Law Poses Key Test for Biden: Protecting Americans from surprise medical bills without inflating health insurance costs will be a pivotal first-year challenge for the Biden administration, industry observers say. The U.S. government will next year create a sweeping system to stop instances where insured individuals get unexpectedly expensive medical bills and to settle pricing disputes between doctors and insurers via arbitration. President Donald Trump signed the legislation into law as part of the omnibus spending and virus relief package. Read more from Alex Ruoff and Danielle Parnass.
More Headlines:
- U.S. Loses Bid to Escape Gilead Claims Over Anti-HIV Drug Patent
- Dept. of Defense Seeks Contractors to Maintain Health IT System
- Drug Firms Must Give Discounts to Contracting Pharmacies: HHS
- New Medicare Drug E-Approval Seeks to Expedite Pickup Process
- N.Y. Medicaid Agency’s Eligibility Remand Practice Wins Approval
To contact the reporters on this story: Brandon Lee in Washington at blee@bgov.com; Jack Fitzpatrick in Washington at jfitzpatrick@bgov.com; Alex Ruoff in Washington at aruoff@bgov.com
To contact the editors responsible for this story: Zachary Sherwood at zsherwood@bgov.com; Giuseppe Macri at gmacri@bgov.com; Michaela Ross at mross@bgov.com
Stay informed with more news like this – from the largest team of reporters on Capitol Hill – subscribe to Bloomberg Government today. Learn more.