HEALTH CARE BRIEFING: Biden Says U.S. Could Send 1 Billion Doses
Bloomberg Government subscribers get the stories like this first. Act now and gain unlimited access to everything you need to know. Learn more.
President Joe Biden said yesterday the U.S. might be able to provide 1 billion more doses of coronavirus vaccine to the world by 2023, and again raised the possibility that the virus may have escaped from a Chinese laboratory.
“There’s a possibility over in 2022, going into 2023, that we would be able to be in a position to provide another billion U.S., but that’s not done yet,” he said yesterday at a news conference in Cornwall, England, following the completion of the Group of Seven summit.
At the meeting, the U.S. promised to send 500 million doses to poorer countries by the end of next year, a move that comes as U.S. demand for vaccines is waning as more of the population is already vaccinated. “There was a clear consensus among all of our colleagues in the G-7 that this wasn’t the end,” he added.
Biden also noted that China has not granted access to its laboratories so scientists can fully study the origins of the Covid-19 pandemic. “Our intelligence community is not certain yet whether or not this was a consequence of” an “experiment gone awry,” he said. “It’s important to know the answer to that.”
Covid-19 loomed large throughout the G-7 meetings, which were teed up by the president announcing a pledge to donate 500 million Pfizer doses to low-income countries, beginning in August and running through 2022. The U.S. contribution marks half of the 1 billion doses G-7 nations pledged at the summit to provide to the rest of the world.
The U.S. doses are also on top of 80 million Biden previously said he would donate. Vaccine makers Pfizer and Moderna have also begun exporting from U.S. facilities directly. Read more from Josh Wingrove and Jennifer Jacobs.
- German Chancellor Angela Merkel said the G-7 would collectively distribute 2.3 billion vaccine doses to developing countries by next year. But Merkel’s figure includes a much wider array of contributions already offered, as well future exports, according to a European official.
- So far, the G-7 nations have pledged only up to 700 million truly new shots, funded in part by previously announced aid. If doses already announced in recent weeks by G-7 and EU nations are included, the number grows to 870 million, as the communique says. Wingrove and Alberto Nardelli have more.
Happening on the Hill
Hearings on the Hill:
- Nominations: The Senate Health, Education, Labor and Pensions Committee meets Wednesday to vote on nominations including Dawn Myers O’Connell to be assistant secretary for preparedness and response, and Miriam E. Delphin-Rittmon to be assistant secretary for mental health and substance use, both for the Health and Human Services Department.
- HHS Policies: The House Education and Labor Committee plans a Wednesday hearing with Health and Human Services Secretary Xavier Becerra on the agency’s policies and priorities.
- Vaccine Legislation: The House Energy and Commerce Health Subcommittee will meet tomorrow to discuss ways to enhance public heath through vaccine legislation.
- Women’s Health Protection Bill: The Senate Judiciary Subcommittee on Constitution plans a hearing Wednesday on the Women’s Health Protection Act.
- Mental Health Care: The Senate Finance Committee meets for a hearing tomorrow on mental health care.
- Care Workers: The Senate Special Committee on Aging holds a hearing Thursday on care workers, family caregivers, seniors and people with disabilities.
- Virus & Student Safety: The Senate Health, Education, Labor & Pensions Committee holds a hearing Thursday on Covid-19 and higher education students returning to campus safely.
Health Bills on Tap: The House this week plans to consider health-care measures under expedited procedure that requires a two-thirds majority for passage:
- VA Podiatrists: The position of podiatric medical director would be created in the Veterans Affairs Department by H.R. 2545. The position would have to be filled by a qualified podiatrist who would report directly to the undersecretary of health. Read the BGOV Bill Summary by Adam M. Taylor.
- VA Hospital Hiring Guidance: The Veterans Affairs Department would have to establish qualifications and performance metrics for Veterans Health Administration human resources positions under H.R. 293. Read the BGOV Bill Summary by Brittney Washington.
- Veterans Access to Contraception: Female veterans wouldn’t be charged copays for contraception under H.R. 239. The Affordable Care Act requires most health insurers to cover contraception without cost-sharing. Under the bill, the same rule would apply to the Veterans Affairs Department. Read the BGOV Bill Summary by Brittney Washington.
Senators Push for Pandemic Prep Commission: Sens. Bob Menendez (D-N.J.) and Susan Collins (R-Maine) are calling for a Sept. 11-style commission to investigate the vulnerabilities of the U.S. public health system in the wake of the coronavirus pandemic, according to an editorial in today’s New York Times. Legislation from the senators would crate “an independent, nonpartisan commission to assess the U.S. pandemic response,” the wrote. “It will illuminate our successes and uncover our failures, and it will provide Congress with actionable recommendations to improve our readiness and prevent future public health crises and the massive loss of life we have suffered from Covid-19.”
Bill Targets Doctors’ Mental Health: Even before the pandemic struck, doctors suffered the worst suicide rate of all professions. Yet many physicians and health-care workers won’t seek counseling or other behavioral health services, fearing it could jeopardize their medical licenses or impede their careers. Lawmakers and advocates are hoping a bill (S. 610) could help change that. Read more from Tony Pugh.
House Eases Mask Rule: Fully vaccinated House lawmakers, staff and visitors will no longer need to use masks anywhere in the Capitol, including committee rooms or the House floor, according to guidance Friday from the Attending Physician Brian Monahan. Unvaccinated or not-fully vaccinated individuals still have to wear a mask, and enforcement is based upon an honor system. Emily Wilkins has more.
The Coronavirus Pandemic
U.S. Death Toll Nears 600,000: The U.S. is approaching 600,000 deaths from Covid, even as new fatalities, infections and hospitalizations continue to plummet nationwide. The vaccination drive has slowed significantly but the results are clear: Masks are disappearing, the country is largely reopening. And daily deaths have fallen from a peak of more than 5,000 in February to an average of about 300 over the last week. Bloomberg News has more updates in the daily virus tracker.
Millions of J&J Doses Cleared for Use: Millions of shots of the Johnson & Johnson Covid-19 vaccine were cleared for use after a months-long delay due to problems at a manufacturer hired to produce the shot, but millions more may end up being thrown away. The Food and Drug Administration released 10 million doses of the vaccine produced by Emergent BioSolutions, people familiar with the matter said. Read more from Riley Griffin and Josh Wingrove.
Related:
- J&J Delivery to South Africa in Limbo After U.S. Ruling
- South Africa Won’t Release Unsuitable Batches of J&J
EEOC’s Vaccine Guidance Leaves Firms Guessing: The U.S. Equal Employment Opportunity Commission guidance on whether incentives can be floated to help encourage workers’ Covid-19 vaccinations raised as many questions as it tried to answered, leaving employers to grapple with what perks may break the law. The workplace agency updated guidance to say firms may offer incentives if they’re not “so substantial as to be coercive.” But what qualifies as coercive remains an open question. Read more from Paige Smith.
- A federal judge has tossed out a lawsuit brought by employees of a Houston hospital contesting a requirement that staff be vaccinated against Covid-19. U.S. District Judge Lynn Hughes of Houston dismissed the suit Saturday in a scathing ruling: He said the claim from 117 employees of the Houston Methodist Hospital that the requirement amounted to Nazi-era human experimentation was both irrelevant and offensive. Read more from Ian Fisher.
Rural Clinics Get $425 Million for Testing: Rural health clinics will receive almost $425 million in funds from the Health and Human Services Department to improve Covid-19 testing and mitigation, the HHS told Bloomberg Law Friday. The funding will be aimed at boosting Covid-19 testing and widening access for rural residents. The funding came from the American Rescue Plan Act, which was signed into law March 11. Read more from Shira Stein.
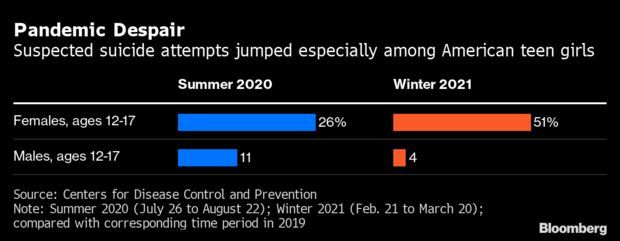
CDC Says Teen Suicide Attempts Surged: Emergency-room visits for suspected suicide attempts by teenagers, particularly girls, rose sharply during coronavirus lockdowns, according to new data from the CDC. The study found that suspected suicide attempts in the 12-17 age group in winter of 2020-2021 by teenaged girls surged to 51% higher than the same period of 2019. Read more from Alex Tanzi.
More U.S. Headlines:
- Daily U.S. Air Travelers Top 2 Million for First Time in Pandemic
- No Causal Association of Myocarditis With Shot, Moderna Says
- Health Providers Get More Time to Report Covid Aid Payments
- California Gov. Newsom Starts Rolling Back Emergency Orders
- Biden Supports Suga in Moving Forward With Olympics: Nikkei
U.S. Presses China on New Covid-19 Study: Secretary of State Antony Blinken pressed his Chinese counterpart in a phone call Friday to allow an expert-led study into the origins of Covid-19, but China’s top diplomat said theories that the virus leaked from a laboratory in Wuhan are absurd, according to readouts of the call. The call was the first time the two diplomats had spoken since a meeting in Alaska in March that got off to a rocky start. Read more from Bloomberg News.
More Global Headlines:
- Johnson Hints at Delay to End of Lockdown as U.K. Cases Spike
- Russia Ups 2020 Covid-19 Death Toll By 38% in Grim Admission
- South African President Urges G7 to Bridge Covid Funding Gap
- Indonesia Races to Vaccinate 1 Million Daily as Infections Spike
- Hot-Spot Paraguay Fights Vaccine Cheaters, Publishing Names
- Saudi Arabia Bars Foreign Hajj Pilgrims in Second Straight Year
What Else to Know Today
Regulatory Agenda Seeks More Active Government: Biden laid out his first regulatory to-do list on Friday, detailing his ambitions to dramatically expand the scope of the government’s involvement in health care. The list, typically issued twice per year, marks a stark departure from the Trump administration’s focus on reducing the size, cost, and scope of U.S. regulations.
- The Centers for Disease Control and Prevention intends to finalize rules by April 2022 for airlines to collect and provide information on communicable diseases. Both the Trump and Obama administrations failed to establish a national aviation preparedness plan for disease outbreak.
- Biden’s HHS will propose repealing a provision in a Trump-era rule that allows states to use brokers and health insurers to directly enroll consumers in Affordable Care Act plans. Such a provision would largely bypass the federal HealthCare.gov exchange site.
- HHS intends to strengthen protections against race, gender and disability discrimination in health care. Courtney Rozen and Dean Scott have more.
- The administration is forging ahead with a divisive, Trump-era regulatory proposal that would limit the government’s ability to control prices on drugs developed from federally funded research. The National Institute of Standards and Technology set an October deadline to update the Bayh-Dole Act, the 1980 law that enables universities to retain patent rights on inventions discovered from government-funded research. The proposal would clarify the law—which allows the government to step in and license federally funded inventions to third parties in certain circumstances—can’t be exercised by an agency primarily to lower costs. Read more from Susan Decker and Ian Lopez.
Trump-Era Insurance Order Appeal Said Moot: Opponents of former President Donald Trump’s order barring immigration by people who lack approved health coverage or the resources to pay for their medical needs asked the Ninth Circuit to toss out the case as moot. Biden May 14 revoked the executive order on which this lawsuit was based, the plaintiffs said. Read more from Mary Anne Pazanowski.
More Headlines:
- Nonprofit’s Challenge to California DNA Rule Partially Revived
- Mental Health Patients Oppose Ending Hospital Boarding Suit
- Biotech Company and Founders Hit With SEC Fraud Allegation
- Lupin Receives Warning Letter From FDA for Somerset Facility
- Ascendis FDA User Fee Goal on Hormone Treatment Extended
To contact the reporter on this story: Brandon Lee in Washington at blee@bgov.com
To contact the editors responsible for this story: Zachary Sherwood at zsherwood@bgov.com; Giuseppe Macri at gmacri@bgov.com; Michaela Ross at mross@bgov.com
Stay informed with more news like this – from the largest team of reporters on Capitol Hill – subscribe to Bloomberg Government today. Learn more.