HEALTH CARE BRIEFING: Biden HHS Pick Faces Senate Fight
Bloomberg Government subscribers get the stories like this first. Act now and gain unlimited access to everything you need to know. Learn more.
President-elect Joe Biden’s pick of California Attorney General Xavier Becerra to lead the Health and Human Services department gives him a fierce defender of Democratic causes but one without hands-on experience leading a massive federal agency in the middle of an unprecedented public health crisis.
Biden satisfied calls for him to name the first Latino to head HHS in keeping with his promise for the most diverse cabinet ever. Unions and Democrats applauded his choice of Becerra, who led challenges to many of President Donald Trump’s efforts to loosen environmental and other regulations and limit immigration and access to health care.
But that may not be enough direct experience to satisfy the GOP-led Senate, which must confirm his nomination.
”I’m not sure what his Health and Human Services credentials are,” Sen. John Cornyn (R-Texas) said. “It’s not like Alex Azar who used to work for pharma and have a health-care background,” he said referring to the current HHS secretary.
Senator Tom Cotton (R-Ark.) called Becerra a “disaster” who was unqualified to lead the agency, but focused his complaints on Becerra’s support for abortion rights.
While he may not have worked in the health industry, Biden found in Becerra someone who does have experience in his other key health-care ambition — to expand the Affordable Care Act. Becerra is an ardent backer of it, having helped write the law and then defended it in court, despite his personal preference for the Medicare for All plan that Biden rejected as a candidate.
Biden plans to introduce his health team today. Read more from Jeffrey Taylor and Billy House.
Biden to Intro Health Team, Braun Says He’ll Meet with Becerra: Biden will hold an event with some of the appointees today in Delaware. Key roles remains unfilled, including commissioner of the Food and Drug Administration and the administrator of the Centers for Medicare and Medicaid Services. It’s not clear when he will name those positions.
Becerra may face difficult Senate confirmations if Republicans keep control of the chamber after two runoff elections in Georgia on Jan. 5. Sen. Mike Braun (R-Ind.), who sits on the Senate health committee, said yesterday he has “serious concerns” about Becerra because of donations he’s received from the health sector, and his support for abortion rights, but that he will meet with him. Braun stopped short of saying he’d vote against Becerra. Read more from Jennifer Epstein, Josh Wingrove, and Shira Stein.
- Defending and expanding Obamacare will be a main priority for Biden, who’s sending a strong message he wants to bolster the law by picking Becerra. The hard-hitting lawyer led the fight to save former President Barack Obama’s signature health-care law in court. If it survives its current challenge in the U.S. Supreme Court, Becerra will run the implementation of the Affordable Care Act, said Katie Keith, a law professor of health at Georgetown University. Read more from Shira Stein and Lydia Wheeler.
Murthy Viewed as Unlikely to Join Cabinet: Vivek Murthy is unlikely to be added to Biden’s cabinet, senior members of the Biden transition team told leaders of the Congressional Asian Pacific American Caucus, two people familiar with the talks said. Caucus leader Judy Chu (D-Calif.) had pushed to get Murthy, who Biden has tapped to be surgeon general, onto the cabinet. But she and others were told Biden is unlikely to make that move in a conference call with Steve Ricchetti, a counselor to the president-elect, Jeffrey Zients, who will coordinate Biden’s coronavirus response, and Ted Kaufman, a transition team leader, Alex Ruoff reports.
US Says Will Meet Vaccine Need Despite Pfizer Report
U.S. officials insisted they’ll have enough Covid-19 vaccine doses to let most Americans get inoculated by next summer, downplaying reports last night that they passed up a chance to secure more of Pfizer’s shot.
The Trump administration is confident that the U.S. will have enough supply to vaccinate everyone, a senior administration official said yesterday on a call with reporters.
Pfizer has told the Trump administration that the U.S. would have to wait until June or July to get more of its vaccine, since it’s done deals with other countries and must deliver doses to them first, the Washington Post reported yesterday. Pfizer offered to sell the government 200 million doses last summer, but the administration opted for half of that, the Post said. The senior administration official rejected the idea that a deal of that size was discussed. Read more from Riley Griffin.
Trump to Hold Vaccine Summit Without Moderna, Pfizer: President Donald Trump plans to hold a “vaccine summit” at the White House today that’s intended to boost confidence in the vaccines among the American public. Trump will open the three-hour indoor summit with remarks, followed by discussions of how the shots were developed, how they’ll be regulated, and how they’ll be administered. He intends to sign an order for HHS to prioritize Americans before helping other countries in need of the medicine. But vaccine producers Moderna and Pfizer, which developed the current leading candidates, won’t be a part of the summit. Read more from Jennifer Jacobs.
States Estimating Vaccines With Outdated Figures: States including New York and Nevada have used outdated federal estimates showing many more doses of Pfizer’s vaccine would be available as they planned for the initial inflow of doses, according to a spokesperson for HHS. The outdated estimates given to states in October suggested there would be 20 million doses of Pfizer’s and BioNTech’s vaccine available, with another 15 million doses from Moderna, the spokesperson said. But recently, federal officials say 6.4 million doses of the Pfizer vaccine and 12.5 million doses of the Moderna shots will be available in the first shipments. John Tozzi and Angelica LaVito have more.
Happening on the Hill
McConnell Refuses to Endorse Bipartisan Stimulus, Risking Deal: Almost a week after Democratic congressional leaders climbed down from their demand for a multi-trillion dollar stimulus package, Senate Majority Leader Mitch McConnell (R-Ky.) continued to tout his own plan, endangering prospects for a compromise. McConnell’s top priority — federal limits on Covid-19 related lawsuits against businesses — has emerged as the key potential deal-breaker. Republicans have balked at the six-month moratorium proposed in a bipartisan stimulus package, saying it’s too limited, and talks have stalled.
McConnell’s continued use of rhetoric that pre-dates the shift by House Speaker Nancy Pelosi (D-Calif.) and Senate Minority Leader Chuck Schumer (D-N.Y.) on the overall dollar amount of aid underscored the risk of no further Covid-19 help by year-end. Read more from Erik Wasson and Laura Litvan.
House Floor: The House today is scheduled to consider several health-related bills, including:
- Medicare Secondary Payments: The Centers for Medicare and Medicaid Services would have to tell liability insurers and workers’ compensation plans when a claimant is enrolled in certain Medicare plans under a modified version of H.R. 1375. The bill would apply when Medicare is the secondary payer for those beneficiaries and a third party requests the information. Rep. Ron Kind (D-Wis.) introduced the bill on Feb. 26, 2019.
- Medicare Enrollment & Other Changes: Enrollment procedures for Medicare beneficiaries would be modified by an expanded version of H.R. 2477, which would eliminate some coverage gaps depending on when a beneficiary enrolls. The expanded bill also would remove a 36-month limit on Medicare coverage following a kidney transplant for immunosuppressive drugs, increase penalties on hospice programs that don’t comply with Medicare standards, and require CMS to share information with certain third parties that are primary payers for enrollees. The House Energy and Commerce Committee approved a version of H.R. 2477 with only the enrollment provisions by voice vote on July 15.
Abortion Cost: The House Appropriations Labor-HHS-Education Subcommittee scheduled a hearing on women being denied abortions due to the cost. House Democratic appropriators including Subcommittee Chairwoman Rosa DeLauro (D-Conn.) have said they want to end the inclusion of the “Hyde amendment” in spending bills. The longstanding provision bars federal funds for abortions except in the case of rape, incest or to save the woman’s life.
- Meanwhile, the Trump administration told the U.S. Supreme Court that the federal rule prohibiting federally funded family planning service providers from referring patients for abortion should get a hearing. A circuit split exists on the question of the rule’s validity that the Supreme Court must resolve, Health and Human Services Secretary Alex Azar said. Read more from Mary Anne Pazanowski.
Covid-19 Outpatient Treatment: The Senate Homeland Security and Governmental Affairs Committee scheduled a hearing on Covid-19 early outpatient treatment.
Senate Floor: The Senate last night by unanimous consent passed an amended version of H.R. 1503, the Orange Book Transparency Act which would amend the Federal Food, Drug, and Cosmetic Act, Nancy Ognanovich and Alex Ruoff report. Senate Health, Education, Labor and Pensions Chair Lamar Alexander (R-Tenn.) introduced the legislation’s substitute amendment, which would reconcile the differences between H.R. 1503 and Section 202 of S. 1895, which was related legislation, among other provisions, according to his aide.
The legislation tried to improve a Food and Drug Administration database of drug approvals and patents, often used to check the availability of generic medicines. H.R. 1503 passed the House in May 2019.
It’s main sponsor, Rep. Robin Kelly (D-Ill.) has said it would bolster competition among drugmakers. The Congressional Budget Office didn’t score it as saving money or having a significant impact on approvals or the availability of new generics.
More on the Pandemic
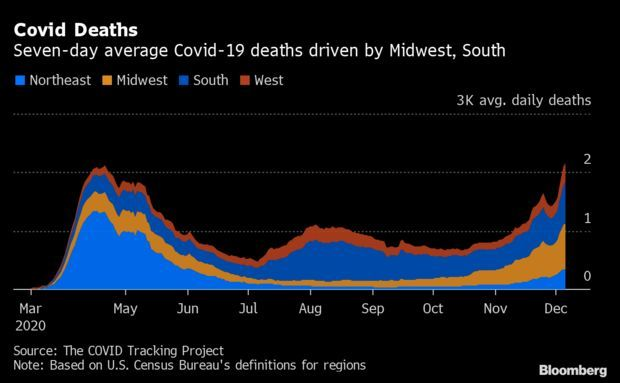
Deaths Match April Peak With Hospitals Still Filling Up: The U.S. is now averaging about as many deaths per day from Covid-19 as it was in April when the pandemic ambushed the New York area. The seven-day average of reported fatalities rose to 2,201 on Sunday, just one shy of the peak on April 18, according to Johns Hopkins University data. Average daily deaths had been predicted in the range of 1,643 to 1,886 through the day after Christmas, according to a month-long forecast from the University of Massachusetts Amherst. Jonathan Levin has more.
Covid-19 Tracking in Schools Key to Reopening: Educators itching to get students back in K-12 classrooms safely say the Biden administration could assist those efforts without Congress by tracking data on the spread of the coronavirus in schools. Such a federal effort would contrast with the Trump administration’s hands-off, decentralized approach, which has urged schools to reopen for in-person instruction but hasn’t tracked how the disease spreads on campuses. Read more from Andrew Kreighbaum.
More Headlines:
- Lower Covid-19 Deaths, Infections Earn Nursing Homes $523 Million
- Pregnant Workers Turn to Courts Over ‘Impossible’ Covid-19 Dilemma
- Workers Administering Covid-19 Vaccines Get CDC Safety Guidance
- Fauci Warns of ‘Dark January’ Amid N.Y. Hospital Capacity Worry
What Else to Know
States Risk Losing Power on Pharmacy Drug Middlemen: States are going to have a harder time controlling the cost of prescription drugs if the Supreme Court widens a federal law that bars states from regulating employee benefit plans. A challenge to an Arkansas law that aims to protect independent pharmacies from the abusive reimbursement practices of rate-setting pharmacy middlemen is testing the bounds of the Employee Retirement Income Security Act. A decision striking down Arkansas’s law could cripple state efforts to control the cost of drugs and other health-care services. Read more from Lydia Wheeler.
More Headlines:
- U.S. Prods High Court to Hear Battle Over Family Planning Rule
- Humana’s Glumetza Suit Against Bausch Is Trimmed, Criticized
- Study Says EPA Model May Misjudge Air Pollution’s Health Cost
- Court Rejects Wholesale Drug Distributor in Accreditation Battle
- U.S. Awards Palantir OK to Power Drug Reviews and Inspections
- CMS Needs More Information on States’ Financing to Improve Oversight (GAO)
With assistance from Alex Ruoff
To contact the reporter on this story: Brandon Lee in Washington at blee@bgov.com
To contact the editors responsible for this story: Zachary Sherwood at zsherwood@bgov.com; Giuseppe Macri at gmacri@bgov.com; Michaela Ross at mross@bgov.com
Stay informed with more news like this – from the largest team of reporters on Capitol Hill – subscribe to Bloomberg Government today. Learn more.