HEALTH CARE BRIEFING: Abortion Riders Likely to Stay in Omnibus
Bloomberg Government subscribers get the stories like this first. Act now and gain unlimited access to everything you need to know. Learn more.
Democrats are nearing defeat on their effort to eliminate longstanding amendments that limit federal funds for abortion in the next government funding package—even after liberals spent years trying to end those restrictions.
Democratic leaders vowed to end language restricting funding for abortion and needle aid when they took control of the House, Senate, and White House in 2021. But senators say they will have to give up on that promise due to the reality of their slim majority in the Senate and rushing to find agreement on a 12-bill appropriations package ahead of the next funding deadline on March 11.
“It’s a 50-50 Senate: if you want to change riders, get 60 people,” said Sen. Jon Tester (D-Mont.), a member of the Senate Appropriations Committee.
Two abortion restrictions — the so-called Hyde and Weldon amendments — have been added to funding bills for the departments of Labor and Health and Human Services for several decades. The Hyde amendment bars federal funds for abortion, except in cases of rape, incest, or to save the life of the woman. The Weldon amendment bars funding for state and local governments if those entities refuse to work with health insurance plans or professionals that refuse to provide, pay for, cover, or refer abortions.
Joe Biden pledged to scrap the Hyde amendment while campaigning for president. Failing to fulfill those promises could hurt Democrats’ chances with voters.
Sen. Roy Blunt (Mo.), the top Republican on the panel responsible for nearly all federal health funds, said he expects the omnibus to retain all the “legacy riders,” or policy provisions that get attached to a must-pass measure to fund the government. He said Democrats simply don’t have the votes to try to repeal them, despite their best efforts to do so. Read more from Alex Ruoff.
Also on Lawmakers’ Radars
Funding Deadline Nears as Lawmakers Debate Vaccine Mandate, Crack Pipes: The Senate today will resume consideration of a House-passed continuing resolution (H.R. 6617) before lawmakers leave town for the Presidents’ Day recess, a move that will avoid a government shutdown and buy time for appropriators in both chambers to finish work on the 12-bill omnibus spending package for fiscal year 2022.
While the GOP supports the measure, lawmakers said they also want votes on amendments that include challenging the Biden administration’s vaccine mandate. Republicans are also demanding a vote on a new bill to prohibit the federal government from purchasing, supplying, or distributing crack pipes or similar drug paraphernalia.
Sen. Marco Rubio (R-Fla.) and many other Republicans also are pushing for a separate vote on the Cutting Rampant Access to Crack Kits (CRACK) Act, which lawmakers recently introduced after they said the Department of Health and Human Services planned to use a $30 million grant program for safe smoking kits that include crack pipes. The Biden administration quickly pushed back, with HHS saying the program to help drug users doesn’t include taxpayer funding to include such pipes in the kits, Nancy Ognanovich reports.
Bill Would Make Social Media Liable for Harm to Kids: Social media platforms would be liable for any harm to minors under a new bipartisan bill unveiled in the Senate yesterday. Content-targeting, unlimited screen time, and engagement-driving features like auto-play also would be disabled by default for minors under a bill offered by Sens. Richard Blumenthal (D-Conn.) and Marsha Blackburn (R-Tenn.). The platforms would also be required to disclose more data to researchers and allow parents to control privacy and account settings. Read more from Brody Ford.
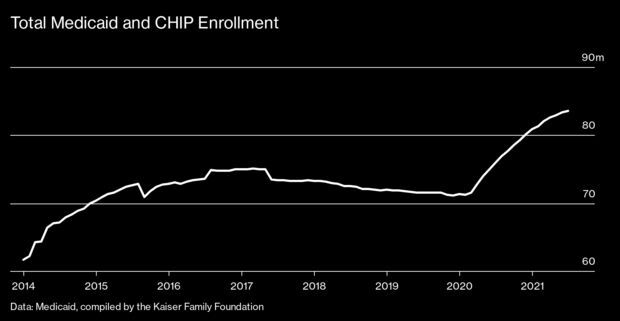
Renewed Eligibility Checks Threaten Medicaid for Millions: At the start of the pandemic, Congress sent extra money to states to bolster their Medicaid programs on the condition that they suspend eligibility reviews for the safety net insurance program. Low-income Americans, the thinking went, risked losing coverage because of bureaucratic snafus just when they needed it most. That agreement—which was extended eight times—is set to expire in April, threatening to leave millions uninsured. Enrollment in Medicaid and the related Children’s Health Insurance Program increased more than 15% from 2019 to mid-2021, to nearly 84 million, federal data show. Anyone with coverage will face a renewal review to make sure they still meet the program’s income requirements and other rules, which vary by state.
Verifying eligibility can be a slog in the best of times, when small discrepancies in a name or address can lead to a loss of coverage. Doing it en masse after a two-year suspension of reviews promises to be even more chaotic. States can try to determine whether residents receiving Medicaid remain eligible using existing data from, say, tax filings or unemployment claims. But if that fails, they’ll need to contact people directly to request further information. That’s where things often get messy. Read more from John Tozzi and Alex Ruoff.
The Coronavirus Pandemic
States Break Ranks on Pandemic Rules: Two years into a global pandemic, states and cities are struggling to answer a critical question: How do you know when to return to normal life? In recent weeks, confusion has played out in particular over masking. A dip in Covid-19 infections and hospitalizations prompted states with some of the most stringent mask rules, including New York and California, to drop their mandates. At the same time, National Institute of Allergy and Infectious Diseases Director Anthony Fauci and U.S. Centers for Disease Control and Prevention Director Rochelle Walensky have both continued to urge caution. Walensky yesterday said the CDC isn’t ready to update its masking guidance, but White House Covid-19 czar Jeff Zients said to expect more information in coming weeks. Madison Muller has more.
Yellen Wants World Bank Fund to Fight Future Pandemics: Treasury Secretary Janet Yellen urged her counterparts from leading industrialized countries to support the establishment of a new World Bank fund intended to prevent and prepare for future global health crises. A new “financial intermediary fund” under the auspices of the World Bank would help address gaps in preparedness, particularly among low-income countries, Yellen said, according to prepared remarks she’s scheduled to deliver virtually today to a meeting of finance ministers and central bank governors from Group of 20 countries. Read more from Christopher Condon.
Texas Sues to Block Biden Mask Mandate: Texas sued to block the Biden administration’s mask mandate for all U.S. travel on airplanes, trains and buses, saying the CDC had exceeded its authority with a rule intended to limit the spread of Covid-19. Texas was joined in filing the complaint by Rep. Elizabeth Van Duyne, who represents the Dallas-Fort Worth area, who was one of three Republicans fined $500 last June for refusing to wear a mask while on the floor of the U.S. House of Representatives. Read more from Laurel Brubaker Calkins.
Mental-Health Scars Stay With Survivors Long After Covid-19 Battle: Early Covid-19 survivors were at higher risk of anxiety, depression and a raft of other mental health problems up to a year after their infections, according to a large U.S. study that widens the scope of the pandemic’s economic and societal impact. The findings, based on an analysis of the U.S. Department of Veterans Affairs’ electronic health-care databases, were published yesterday in the journal BMJ. They indicate that the burden of neurological disorders—estimated to afflict more than 200 million Americans in 2017—will balloon as a consequence of the pandemic and have wider medical and social complications, including more substance abuse and suicides. Jason Gale has more.
Pfizer Pill Easier to Get as Omicron Fades: As the omicron wave peaked in the U.S. last month, the first-line treatment for high-risk patients with early Covid-19 dangled out of reach for most. Only a small trickle of the new Paxlovid pill from Pfizer was reaching hospitals and pharmacies. Now as cases plummet nationwide and the drugmaker continues to deliver hundreds of thousands of doses ordered by the U.S. government to pharmacies, Paxlovid is starting to seem plentiful. Read more from Carey Goldberg.
Study Calms Fears of Harsher Omicron Subvariant: The omicron BA.2 subvariant doesn’t cause more severe disease than the original version, according to a South African study that appears to allay fears it causes harsher illness. Patients infected with the new subvariant suffered from a similar rate of severe disease and hospitalization as those infected by the original omicron strain, researchers from the National Institute for Communicable Diseases found in data from almost 100,000 cases. The study indicates that, like the initial version, BA.2 is relatively mild in comparison with earlier strains, like delta. Read more from Antony Sguazzin.
What Else to Know Today
Biden Taps OSTP Temporary Leadership: Biden yesterday tapped Dr. Alondra Nelson to perform the duties of director of the White House Office of Science and Technology Policy and former NIH director Dr. Francis Collins to perform the duties of science advisor to the president until permanent leadership is confirmed. In a statement, the White House said Nelson and Collins will help push forward Biden’s priorities for “the creation of a new ARPA-H research and discovery agency, the building of support for a Cancer Moonshot 2.0, the search for a new head of NIH, and the broad advisory work of PCAST.”
AstraZeneca Wins Bid to Quash HHS’s Drug Discount Demand: The Health and Human Services Department violated procedural rules in demanding that AstraZeneca Pharmaceuticals provide steeply-discounted medicines through a government program embroiled in litigation, a federal judge ruled. The decision yesterday is the latest turn in an ongoing saga between drugmakers and the Biden and Trump administrations over a discount program geared for low-income patients. The U.S. District Court for the District of Delaware vacated and sent back to the HHS a letter notifying AstraZeneca that it must resume discounts it had limited for covered providers that dispense drugs through for-profit pharmacies. Read more from Ian Lopez.
Drug Price Reporting Rules Foiled by Loopholes in Supply Chain: U.S. transparency rules for drug and health costs are being stymied by interlocking business deals between insurers, pharmacy liaisons, and other companies, including some that are overseas. Those are observations gleaned from industry comments about a multi-agency rule (RIN 0938 AU66) designed to give employers and insurers a window into how the care they’re paying for is priced. Entities at every stage of the supply chain say they need more information on what the other companies in the system are paying. The reporting requirements went into effect in December, and regulators are seeking input into how to clarify the rules. Read more from Sara Hansard.
More Headlines:
- Abortion Pill Is Safer Than Tylenol and Almost Impossible to Get
- OptumRx Wins Exit From Antitrust, RICO Case Over Insulin Prices
- Ex-GlaxoSmithKline Employee Barred From Using Trade Secrets
To contact the reporter on this story: Brandon Lee in Washington at blee@bgov.com
To contact the editors responsible for this story: Zachary Sherwood at zsherwood@bgov.com; Giuseppe Macri at gmacri@bgov.com; Michaela Ross at mross@bgov.com
Stay informed with more news like this – from the largest team of reporters on Capitol Hill – subscribe to Bloomberg Government today. Learn more.